Ampakines, also stylized as AMPAkines, are a subgroup of AMPA receptor positive allosteric modulators with a benzamide or closely related chemical structure. They are also known as “CX compounds”. Ampakines take their name from the AMPA receptor (AMPAR), a type of ionotropic glutamate receptor with which the ampakines interact and act as positive allosteric modulators (PAMs) of. Although all ampakines are AMPAR PAMs, not all AMPAR PAMs are ampakines.
They are currently being investigated as potential treatment for a range of conditions involving mental disability and disturbances such as Alzheimer’s disease, Parkinson’s disease, schizophrenia, treatment-resistant depression (TRD) or neurological disorders such as attention deficit hyperactivity disorder (ADHD), among others.
More recently developed ampakine compounds are much more potent and selective for the AMPA receptor target, and while none of the newer selective ampakine compounds have yet come onto the market, various ampakines are in clinical trials.
Effects
Unlike the first stimulants (such as caffeine, methylphenidate (Ritalin) and amphetamines), ampaquine does not evidence unpleasant side effects such as insomnia
They are currently being investigated as a potential treatment for a wide range of conditions involving mental disabilities and disorders such as Alzheimer’s, Parkinson’s disease, schizophrenia, treatment-resistant depression (DRT), or neurological disorders such as deficit disorder of attention with hyperactivity (ADHD), among others.
The activity of ampaquins has been established as the modes of action of the already well-known class of nootropics, racetam drugs, such as piracetam, aniracetam, oxiracetam, and pramiracetam, however, these drugs have multiple modes of action and produce only AMPA weak activation, and it is not clear how significant the actions of ampaquins in the production of nootropic effects are. Recent ampaquine compounds are more potent and selective for AMPA receptor targets, and while none of the selective ampaquine receptors are commercially available, one compound, CX717, is in phase II clinical trials since 2008
Examples and structures
Five structural classes of ampaquine drugs have been developed at a considerable time:
Drugs are derived from pyrrolidine, such as piracetam and aniracetam
The CX drug series encompassing the piperidine and pyrrolidine structures
Drugs derived from thiazide, such as cyclotiazide and IDRA-21
Sulfonamides such as LY-392,098, LY-404,187, LY-451,646, LY-503,430 and Unifiram
Drugs derived from benzylpiperazine, such as sunifiram
Family Racetam
The parent compound, in which the first modulation of AMPA activity was first characterized, was the well-known nootropic aniracetam. Several drugs of the racetam family have been reported as producers of amphetamine effects. Although this behavior is well-established for some compounds, such as aniracetam and pramiracetam, it is still undefined if the entire race family acts in this way, since race drugs appear to have multiple modes of action.
Cortex Pharmaceuticals (pharmaceutical industry)
Since the discovery of the mode of action of ampaquins as one of the means by which racetams produce their nootropic effects, a wide range of amphokinins such as increased selectivity have been developed by Cortex Pharmaceuticals; patents covering most of the medical uses of that class of medicine. The most well-known compounds resulting from Cortex’s drug development program are:
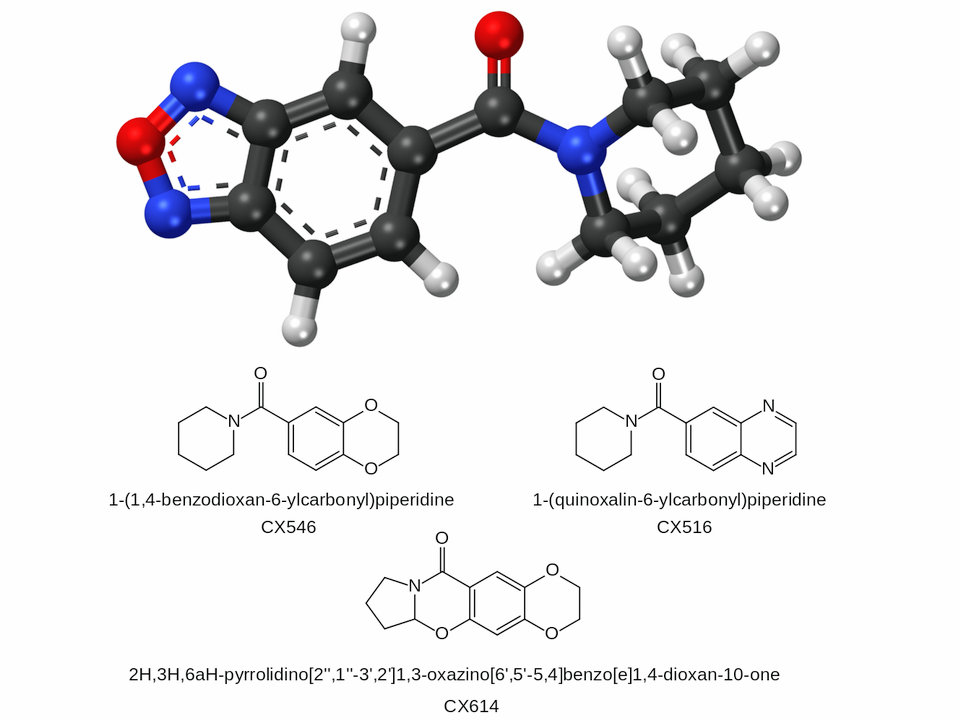
CX-516 (Ampalex)
CX-546
CX-614
CX-691 (Farampator)
CX717
Org. 26576. This drug was invented by Cortex, but it was licensed to Organon for development purposes
Many other compounds, such as CX-701, CX-1739, CX-1763 and CX-1837, are in the early developmental stage, and while little information about them has been released, CX-1739 is believed to be the class so far, with 5x the power of the CX-717
Eli Lilly and Schering-Plow
Other compounds that produce the activity profile of the ampaquins, such as Eli Lilly’s IDRA-21 and LY-503,430, along with uni and sunifiram have been developed by other pharmaceutical companies, but restricted to animal testing to date. Cortex is the only company that currently develops selective tamping machines for human use, in partnership with the large pharmaceutical industry Schering-Plow.
Development
A wide range of ampakines have been developed by RespireRx, which hold patents covering most medical uses of this class of drugs. The best known compounds that have come out of the RespireRx drug development program are CX-516 (Ampalex), CX-546, CX-614, CX-691 (farampator), and CX-717. ORG-26576 was developed by RespireRx but then licensed to Organon for development.
Several other compounds such as CX-701, CX-1739, CX-1763 and CX-1837 have also been announced as being under current investigation, and while little information has yet been released about them, CX-1739 is believed to be the most potent compound in this class to date, reportedly some 5x the potency of CX-717.
Presently, CX717 is in phase II clinical trials as a possible non-stimulant pharmacotherapy in the treatment of ADHD. As the AMPA receptors also mediate respiratory drive, CX717 is also being investigated as a therapy in opioid-induced respiratory depression and spinal-cord injury.
Mechanism of action
Ampakines work by allosterically binding to a type of ionotropic glutamate receptor, called AMPA receptors.
The ampakines are mostly low-impact AMPAR PAMs, though with some exceptions, such as tulrampator (S-47445, CX-1632).
Side effects
Few side effects have been determined, but an ampakine called farampator (CX-691) has side effects including headache, drowsiness, nausea, and impaired episodic memory.
Medical applications
An ampakine called CX456 has been proposed as a treatment for Rett syndrome, after favorable testing in an animal model.
Ampakines have been investigated by DARPA for potential use in increasing military effectiveness.
Source from Wikipedia