A printable organ is an artificially constructed device designed for organ replacement, produced using 3D printing techniques. The primary purpose of printable organs is in transplantation. Research is currently being conducted on artificial heart, kidney, and liver structures, as well as other major organs. For more complicated organs, such as the heart, smaller constructs such as heart valves have also been the subject of research. Some printed organs are approaching functionality requirements for clinical implementation, and primarily include hollow structures such as the bladder, as well as vascular structures such as urine tubes.
3D printing allows for the layer-by-layer construction of a particular organ structure to form a cell scaffold. This can be followed by the process of cell seeding, in which cells of interest are pipetted directly onto the scaffold structure. Additionally, the process of integrating cells into the printable material itself, instead of performing seeding afterwards, has been explored.
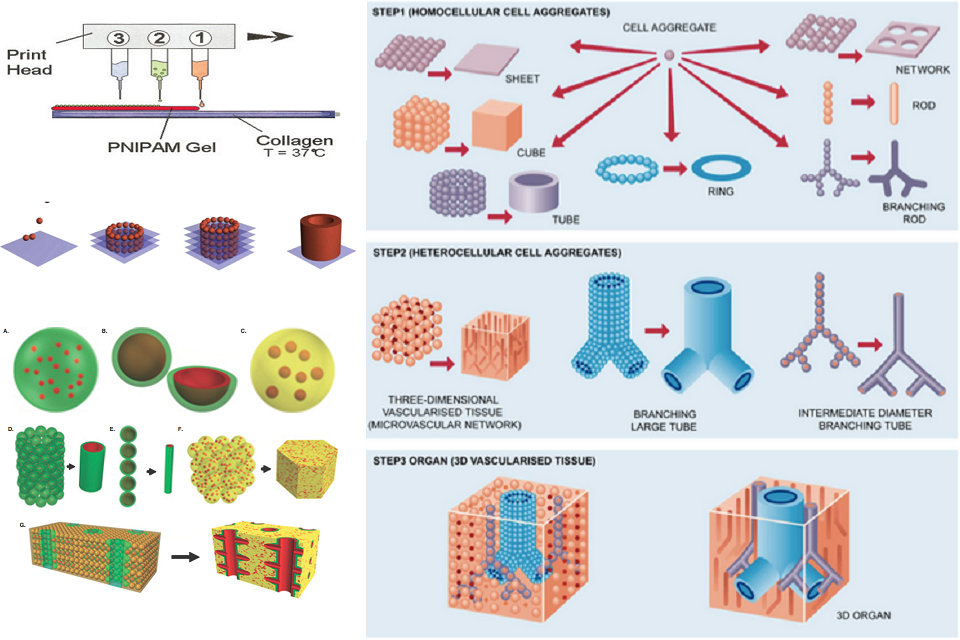
Modified inkjet printers have been used to produce three-dimensional biological tissue. Printer cartridges are filled with a suspension of living cells and a smart gel, the latter used for providing structure. Alternating patterns of the smart gel and living cells are printed using a standard print nozzle, with cells eventually fusing together to form tissue. When completed, the gel is cooled and washed away, leaving behind only live cells.
History
3D printing for producing a cellular construct was first introduced in 2003, when Thomas Boland of Clemson University patented the use of inkjet printing for cells. This process utilized a modified spotting system for the deposition of cells into organized 3D matrices placed on a substrate.
Since Boland’s initial findings, the 3D printing of biological structures, also known as bioprinting, has been further developed to encompass the production of tissue and organ structures, as opposed to cell matrices. Additionally, more techniques for printing, such as extrusion bioprinting, have been researched and subsequently introduced as a means of production.
Organ printing has been approached as a potential solution for the global shortage of donor organs. Organs that have been successfully printed and implemented in a clinical setting are either flat, such as skin, vascular, such as blood vessels, or hollow, such as the bladder. When artificial organs are prepared for transplantation, they are often produced with the recipient’s own cells.
More complex organs, namely those that consist of solid cellular structures, are undergoing research; these organs include the heart, pancreas, and kidneys. Estimates for when such organs can be introduced as a viable medical treatment vary. In 2013, the company Organovo produced a human liver using 3D bioprinting, though it is not suitable for transplantation, and has primarily been used as a medium for drug testing.
Approaches
Researchers have developed different approaches to producing living synthetic organs. The 3D bio-printing is based on three main approaches: Biomimicry, autonomous self-assembly and construction of mini tissue blocks.
Biomimicry
The first approach to bio-printing is called biomimicry. The main objective of this approach is to create structures identical to natural structures. Biomimicry requires duplication of the shape, frame and micro-environment of organs and tissues. Biomimicry application in bio-printing involves the identical copy of the cellular and extracellular parts of the organs. For this approach to be successful, tissue replication at a micron scale is required. This degree of precision involves understanding the microenvironment, the nature of biological forces, the precise organization of cells, solubility factors and the composition and structure of the extracellular matrix.
Self-assembly
The second approach used in bio-printing is autonomous self-assembly. This approach relies on the natural physical process of developing embryonic organs. When the cells are in their early development phase, they create their own extracellular matrix building block, and produce the proper cell signaling of their own and take the layout and microarchitecture required to provide the expected biological functions. Autonomous self-assembly requires knowledge of the processes of development of tissues and organs in the embryo. Autonomous self-assembly relies on cell capabilities as the fundamental building block of histogenesis. This technique therefore requires a very thorough understanding of the mechanisms of embryonic tissue development as well as the micro-environments in which tissues grow.
Mini-fabric
The third approach to bio-printing is a combination of both biomimetic and self-assembly approaches. This technique is referred to as “mini-tissues”. Organs and tissues are made from very small functional components. The mini-fabric approach is to take these small pieces and arrange them in a larger structure. This approach uses two different strategies. The first strategy is to use self-assembled cell spheres in large scale fabrics, using natural patterns as a guide. The second strategy is to develop accurate reproductions and high quality of fabric and allow them to mount automatically in large functional fabrics to scale. The mixing of these strategies is necessary to print a complex three-dimensional biological structure.
Organ printing has great potential for NBIC technologies (nano, bio, info and cognitive) to advance medicine and surgical procedures, to save time, reduce costs and create new opportunities for patients and patients. health professionals.
3D printing techniques
3D printing for the manufacturing of artificial organs has been a major topic of study in biological engineering. As the rapid manufacturing techniques entailed by 3D printing become increasingly efficient, their applicability in artificial organ synthesis has grown more evident. Some of the primary benefits of 3D printing lie in its capability of mass-producing scaffold structures, as well as the high degree of anatomical precision in scaffold products. This allows for the creation of constructs that more effectively resemble the microstructure of a natural organ or tissue structure.
Organ printing using 3D printing can be conducted using a variety of techniques, each of which confers specific advantages that can be suited to particular types of organ production. Two of the most prominent types of organ printing are drop-based bioprinting and extrusion bioprinting. Numerous other ones do exist, though are not as commonly used, or are still in development.
Drop-based bioprinting (Inkjet)
Drop-based bioprinting creates cellular constructs using individual droplets of a designated material, which has oftentimes been combined with a cell line. Upon contact with the substrate surface, each droplet begins to polymerize, forming a larger structure as individual droplets begin to coalesce. Polymerization is instigated by the presence of calcium ions on the substrate, which diffuse into the liquified bioink and allow for the formation of a solid gel. Drop-based bioprinting is commonly used due to its efficient speed, though this aspect makes it less suitable for more complicated organ structures.
Extrusion bioprinting
Extrusion bioprinting involves the constant deposition of a particular printing material and cell line from an extruder, a type of mobile print head. This tends to be a more controlled and gentler process for material or cell deposition, and allows for greater cell densities to be used in the construction of 3D tissue or organ structures. However, such benefits are set back by the slower printing speeds entailed by this technique. Extrusion bioprinting is often coupled with UV light, which photopolymerizes the printed material to form a more stable, integrated construct.
Printing materials
Materials for 3D printing usually consist of alginate or fibrin polymers that have been integrated with cellular adhesion molecules, which support the physical attachment of cells. Such polymers are specifically designed to maintain structural stability and be receptive to cellular integration. The term “bioink” has been used as a broad classification of materials that are compatible with 3D bioprinting.
Printing materials must fit a broad spectrum of criteria, one of the foremost being biocompatibility. The resulting scaffolds formed by 3D printed materials should be physically and chemically appropriate for cell proliferation. Biodegradability is another important factor, and insures that the artificially formed structure can be broken down upon successful transplantation, to be replaced by a completely natural cellular structure. Due to the nature of 3D printing, materials used must be customizable and adaptable, being suited to wide array of cell types and structural conformations.
Hydrogel alginates have emerged as one of the most commonly used materials in organ printing research, as they are highly customizable, and can be fine-tuned to simulate certain mechanical and biological properties characteristic of natural tissue. The ability of hydrogels to be tailored to specific needs allows them to be used as an adaptable scaffold material, that are suited for a variety of tissue or organ structures and physiological conditions. A major challenge in the use of alginate is its stability and slow degradation, which makes it difficult for the artificial gel scaffolding to be broken down and replaced with the implanted cells’ own extracellular matrix. Alginate hydrogel that is suitable for extrusion printing is also often less structurally and mechanically sound; however, this issue can be mediated by the incorporation of other biopolymers, such as nanocellulose, to provide greater stability. The properties of the alginate or mixed-polymer bioink are tunable and can be altered for different applications and types of organs.
Organ structures
While many of the technical challenges of organ printing are shared with other applications of 3D bioprinting, there are some organ-specific structural elements that must be addressed for successful creation of a transplantable printed organ.
Vascularization
The transfer of nutrients and oxygen to cells throughout a printed organ is essential for its function. In very small or thin tissues of less than a millimeter in thickness, cells can receive nutrients through diffusion. However, larger organs require the transportation of nutrients to cells deeper inside the tissue, which requires that the tissue be vascularized, and thus able to receive blood for the exchange of cargo like oxygen and cell wastes. Early organ printing techniques created solid tissues that were unable to vascularize, or vascularized only slowly as host blood vessels entered the transplant, leading to issues like necrosis inside the tissue that can threaten the health and successful recovery of a transplant recipient. More recently developed techniques allow printed organs to be created with a more complex 3D structure, including preexisting internal vasculature, that permits faster integration of the transplant into the host circulatory system. There are multiple techniques for creating vascular systems currently under development. One method is the separate extrusion printing of vessels that are then incorporated into a larger tissue. Another method is sacrificial printing, in which the entire tissue is printed at once, and a dissolvable or otherwise removable bioink is used to form the interior of the vessels. Once this sacrificial scaffolding is removed, usually by a chemical or thermal method, the rest of tissue then contains a vascular pattern.
Cell sources
The creation of a complete organ often requires incorporation of a variety of different cell types, arranged in distinct and patterned ways. One advantage of 3D-printed organs, compared to traditional transplants, is the potential to use cells derived from the patient to make the new organ. This significantly decreases the likelihood of transplant rejection, and may remove the need for immunosuppressive drugs after transplant, which would reduce the health risks of transplants. However, since it may not always be possible to collect all the needed cell types, it may be necessary to collect adult stem cells or induce pluripotency in collected tissue. This involves resource-intensive cell growth and differentiation and comes with its own set of potential health risks, since cell proliferation in a printed organ occurs outside the body and requires external application of growth factors. However, the ability of some tissues to self-organize into differentiated structures may provide a way to simultaneously construct the tissues and form distinct cell populations, improving the efficacy and functionality of organ printing.
Source from Wikipedia