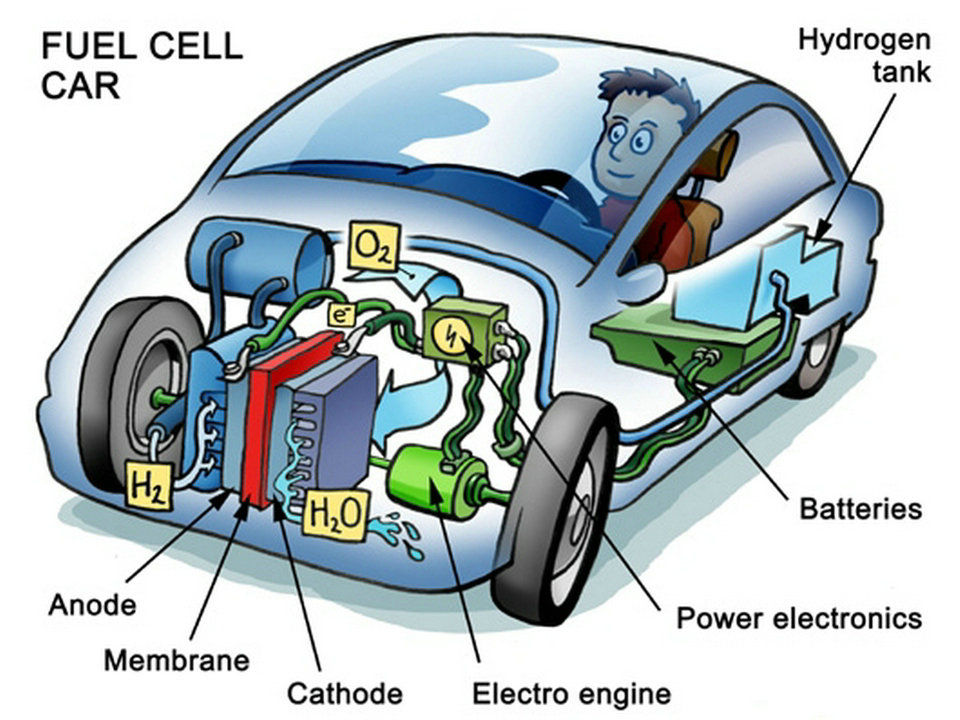
A hydrogen vehicle is a vehicle that uses hydrogen as its onboard fuel for motive power. Hydrogen vehicles include hydrogen-fueled space rockets, as well as automobiles and other transportation vehicles. The power plants of such vehicles convert the chemical energy of hydrogen to mechanical energy either by burning hydrogen in an internal combustion engine, or by reacting hydrogen with oxygen in a fuel cell to run electric motors. Widespread use of hydrogen for fueling transportation is a key element of a proposed hydrogen economy.
As of 2016, there are 3 hydrogen cars publicly available in select markets: the Toyota Mirai, the Hyundai ix35 FCEV, and the Honda Clarity. Several other companies are working to develop hydrogen cars. As of 2014, 95% of hydrogen is made from natural gas. It can be produced using renewable sources, but that is an expensive process. Integrated wind-to-hydrogen (power-to-gas) plants, using electrolysis of water, are exploring technologies to deliver costs low enough, and quantities great enough, to compete with hydrogen production using natural gas. The drawbacks of hydrogen use are high carbon emissions intensity when produced from natural gas, capital cost burden, low energy content per unit volume, production and compression of hydrogen, and the large investment in infrastructure that would be required to fuel vehicles.
Energy carrier hydrogen
Fuel and exhaust gases
The hydrogen used as fuel is not primary energy , but must be produced from primary energy analogous to power generation . To his production is energy required. This is in the chemical reaction in a hydrogen combustion engine or in the fuel cellpartly released again. Due to its low density, hydrogen gas contains more energy per unit weight per unit of mass than any other chemical fuel. However, the energy density is very low by volume. Therefore, hydrogen as fuel must be either highly compressed (up to about 700 bar) or liquefied (-253 ° C). Both are associated with additional energy input.
The exhaust gases of a fuel cell consist of pure water vapor .
During the combustion of hydrogen in combination with air (in a gas turbine), the exhaust gases additionally contain nitrogen oxides , which arise from the atmospheric nitrogen at the high temperatures in the combustion chamber . At high excess air (λ»1) less nitrogen oxides are produced, but then the efficiency also decreases. In piston engines continue to get traces of CO and CH in the exhaust gas. They come from the lubricating oil between the cylinder wall and the piston and from the crankcase breather .
Hydrogen production
The main processes for hydrogen production are
The thermo-chemical conversion of carbon- energy sources (usually Fossil fuels ) at temperatures of 300-1000 ° C . The oldest process of this type is steam reforming with a market share of over 90%. Using this process, the city gas ( synthesis gas) used to be produced from coal and water vapor , which contained approx. 60% hydrogen. Through further process steps, almost the entire energy content of the energy source can be bound to hydrogen. The disadvantage here is the resulting climate-damaging gas CO 2, There are also technologies for making hydrogen carbon-neutral from biomass. A first commercial plant, the Blue Tower Herten , was due to bankruptcy of Solar Millennium AG is not completed.
Hydrogen is a by-product of a number of chemical processes (eg, chlor-alkali electrolysis ). The quantities are considerable, but are mostly reused. The hydrogen produced as a by-product in the Cologne region alone would be sufficient to operate 40,000 cars on a permanent basis (as of 2010).
Still comparatively seldom, hydrogen is produced by the electrolysis of water . Efficiencies of 70-80% are achieved. There are currently projects in which the electrolyser is supplied directly by wind turbines. Wind turbines are now disconnected on windy days with low electricity demand; instead, they could then be used for electrolysis for the production of hydrogen. In addition to the necessary amount of energy, the problem is the provision of the required water:To supply all the aircraft refueling at Frankfurt Airport with hydrogen from the electrolysis of water would require the power of 25 large-scale power plants. At the same time, the water consumption of Frankfurt would double.”
Attempts to produce hydrogen in a hydrogen bioreactor with algae via a variant of photosynthesis are currently still in the research stage.
Hydrogen storage
The technical problems with the storage of hydrogen are considered solved today. Methods such as pressurized and liquid hydrogen storage and storage in metal hydrides are in commercial use. In addition, there are other methods such as storage in nanotubes or as a chemical compound ( N -Ethylcarbazol ), which are still in the development stage or in basic research.
Hydrogen station
A precondition for the widespread use of hydrogen drives is the production of the supply infrastructure. To get a nationwide network in Germany, about 1000 hydrogen refueling stations are required.
There are approximately 274 hydrogen refueling stations worldwide (as of May 2017). In Germany there are about 30, of which only 7 are publicly operated. In cooperation with Linde AG, the Daimler Group will build another 20 hydrogen refueling stations in order to initially ensure continuous connections on the north-south and east-west axes. → See also: Hydrogen highway
A hydrogen filling station costs about 1 to 1.5 million euros.
Vehicles
Automobiles, buses, forklifts, trains, PHB bicycles, canal boats, cargo bikes, golf carts, motorcycles, wheelchairs, ships, airplanes, submarines, and rockets can already run on hydrogen, in various forms. NASA used hydrogen to launch Space Shuttles into space. A working toy model car runs on solar power, using a regenerative fuel cell to store energy in the form of hydrogen and oxygen gas. It can then convert the fuel back into water to release the solar energy. Since the advent of hydraulic fracturing the key concern for hydrogen fuel cell vehicles is consumer and public policy confusion concerning the adoption of natural gas powered hydrogen vehicles with heavy hidden emissions to the detriment of environmentally friendly transportation.
A land-speed record for a hydrogen-powered vehicle of 286.476 miles per hour (461.038 km/h) was set by Ohio State University’s Buckeye Bullet 2, which achieved a “flying-mile” speed of 280.007 miles per hour (450.628 km/h) at the Bonneville Salt Flats in August 2008. A record of 207.297 miles per hour (333.612 km/h) was set by a prototype Ford Fusion Hydrogen 999 Fuel Cell Race Car at the Bonneville Salt Flats, in August 2007, using a large compressed oxygen tank to increase power.
Automobiles
As of 2016, there are 3 hydrogen cars publicly available in select markets: the Toyota Mirai, the Hyundai ix35 FCEV, and the Honda Clarity.
Toyota launched its first production fuel cell vehicle (FCV), the Mirai, in Japan at the end of 2014 and began sales in California, mainly the Los Angeles area, in 2015. The car has a range of 312 mi (502 km) and takes about five minutes to refill its hydrogen tank. The initial sale price in Japan was about 7 million yen ($69,000). Former European Parliament President Pat Cox estimated that Toyota would initially lose about $100,000 on each Mirai sold. Many automobile companies have introduced demonstration models in limited numbers (see List of fuel cell vehicles and List of hydrogen internal combustion engine vehicles). One disadvantage of hydrogen compared to other automobile fuels is its low density.
In 2013 BMW leased hydrogen technology from Toyota, and a group formed by Ford Motor Company, Daimler AG, and Nissan announced a collaboration on hydrogen technology development. By 2017, however, Daimler had abandoned hydrogen vehicle development, and most of the automobile companies developing hydrogen cars had switched their focus to battery electric vehicles.
Buses
Fuel-cell buses (as opposed to hydrogen fueled buses) are being trialed by several manufacturers in different locations, for example, Ursus Lublin. The Fuel Cell Bus Club is a global fuel cell bus testing collaboration.
Trams and trains
In March 2015, China South Rail Corporation (CSR) demonstrated the world’s first hydrogen fuel cell-powered tramcar at an assembly facility in Qingdao. The chief engineer of the CSR subsidiary CSR Sifang Co Ltd., Liang Jianying, said that the company is studying how to reduce the running costs of the tram. Tracks for the new vehicle have been built in seven Chinese cities. China plans to spend 200 billion yuan ($32 billion) through 2020 to increase tram tracks to more than 1,200 miles.
In northern Germany in 2018 the first fuel-cell powered Coradia iLint trains were placed into service; excess power is stored in lithium-ion batteries.
Bicycles
In 2007, Pearl Hydrogen Power Sources of Shanghai, China, unveiled a hydrogen bicycle at the 9th China International Exhibition on Gas Technology, Equipment, and Applications.
Military vehicles
General Motors’ military division, GM Defense, focuses on hydrogen fuel cell vehicles. Its SURUS (Silent Utility Rover Universal Superstructure) is a flexible fuel cell electric platform with autonomous capabilities. Since April 2017, the U.S. Army has been testing the commercial Chevrolet Colorado ZH2 on its U.S. bases to determine the viability of hydrogen-powered vehicles in military mission tactical environments.
Motorcycles and scooters
ENV develops electric motorcycles powered by a hydrogen fuel cell, including the Crosscage and Biplane. Other manufacturers as Vectrix are working on hydrogen scooters. Finally, hydrogen-fuel-cell-electric-hybrid scooters are being made such as the Suzuki Burgman fuel-cell scooter. and the FHybrid. The Burgman received “whole vehicle type” approval in the EU. The Taiwanese company APFCT conducted a live street test with 80 fuel-cell scooters for Taiwan’s Bureau of Energy.
Quads and tractors
Autostudi S.r.l’s H-Due is a hydrogen-powered quad, capable of transporting 1-3 passengers. A concept for a hydrogen-powered tractor has been proposed.
Airplanes
Companies such as Boeing, Lange Aviation, and the German Aerospace Center pursue hydrogen as fuel for manned and unmanned airplanes. In February 2008 Boeing tested a manned flight of a small aircraft powered by a hydrogen fuel cell. Unmanned hydrogen planes have also been tested. For large passenger airplanes, The Times reported that “Boeing said that hydrogen fuel cells were unlikely to power the engines of large passenger jet airplanes but could be used as backup or auxiliary power units onboard.”
In July 2010, Boeing unveiled its hydrogen-powered Phantom Eye UAV, powered by two Ford internal-combustion engines that have been converted to run on hydrogen.
In Britain, the Reaction Engines A2 has been proposed to use the thermodynamic properties of liquid hydrogen to achieve very high speed, long distance (antipodal) flight by burning it in a precooled jet engine.
Fork trucks
A HICE forklift or HICE lift truck is a hydrogen fueled, internal combustion engine-powered industrial forklift truck used for lifting and transporting materials. The first production HICE forklift truck based on the Linde X39 Diesel was presented at an exposition in Hannover on May 27, 2008. It used a 2.0 litre, 43 kW (58 hp) diesel internal combustion engine converted to use hydrogen as a fuel with the use of a compressor and direct injection.
A fuel cell forklift (also called a fuel cell lift truck) is a fuel cell powered industrial forklift truck. In 2013 there were over 4,000 fuel cell forklifts used in material handling in the US. The global market was estimated at 1 million fuel cell powered forklifts per year for 2014–2016. Fleets are being operated by companies around the world. Pike Research stated in 2011 that fuel-cell-powered forklifts will be the largest driver of hydrogen fuel demand by 2020.
Most companies in Europe and the US do not use petroleum powered forklifts, as these vehicles work indoors where emissions must be controlled and instead use electric forklifts. Fuel-cell-powered forklifts can provide benefits over battery powered forklifts as they can be refueled in 3 minutes. They can be used in refrigerated warehouses, as their performance is not degraded by lower temperatures. The fuel cell units are often designed as drop-in replacements.
Rockets
Many large rockets use liquid hydrogen as fuel, with liquid oxygen as an oxidizer (LH2/LOX). An advantage of hydrogen rocket fuel is the high effective exhaust velocity compared to kerosene/LOX or UDMH/NTO engines. According to the Tsiolkovsky rocket equation, a rocket with higher exhaust velocity uses less propellant to accelerate. Also the energy density of hydrogen is greater than any other fuel. LH2/LOX also yields the greatest efficiency in relation to the amount of propellant consumed, of any known rocket propellant.
A disadvantage of LH2/LOX engines is the low density and low temperature of liquid hydrogen, which means bigger and insulated and thus heavier fuel tanks are needed. This increases the rocket’s structural mass which reduces its delta-v significantly. Another disadvantage is the poor storability of LH2/LOX-powered rockets: Due to the constant hydrogen boil-off, the rocket must be fueled shortly before launch, which makes cryogenic engines unsuitable for ICBMs and other rocket applications with the need for short launch preparations.
Overall, the delta-v of a hydrogen stage is typically not much different from that of a dense fuelled stage, but the weight of a hydrogen stage is much less, which makes it particularly effective for upper stages, since they are carried by the lower stages. For first stages, dense fuelled rockets in studies may show a small advantage, due to the smaller vehicle size and lower air drag.
LH2/LOX were also used in the Space Shuttle to run the fuel cells that power the electrical systems. The byproduct of the fuel cell is water, which is used for drinking and other applications that require water in space.
Heavy Truck
In 2016 Nikola Motor Company introduced a hydrogen-powered Class 8 heavy truck powered by a 320 kWh EV battery. Nikola plans two versions of the hydrogen powered truck, long haul Nikola One and day cab Nikola Two. United Parcel Service began testing of a hydrogen powered delivery vehicle in 2017. US Hybrid, Toyota, and Kenworth have also announced plans to test Class 8 drayage hydrogen fuel cell trucks.
Internal combustion vehicle
Hydrogen internal combustion engine cars are different from hydrogen fuel cell cars. The hydrogen internal combustion car is a slightly modified version of the traditional gasoline internal combustion engine car. These hydrogen engines burn fuel in the same manner that gasoline engines do; the main difference is the exhaust product. Gasoline combustion results in carbon dioxide and water vapor, while the only exhaust product of hydrogen combustion is water vapor.
In 1807 Francois Isaac de Rivaz designed the first hydrogen-fueled internal combustion engine. In 1965, Roger Billings, then a high school student, converted a Model A to run on hydrogen. In 1970 Paul Dieges patented a modification to internal combustion engines which allowed a gasoline-powered engine to run on hydrogen US 3844262.
Mazda has developed Wankel engines burning hydrogen. The advantage of using an internal combustion engine, like Wankel and piston engines, is the lower cost of retooling for production.
HICE forklift trucks have been demonstrated based on converted diesel internal combustion engines with direct injection.
Fuel cell
Fuel cell cost
Hydrogen fuel cells are relatively expensive to produce, as their designs require rare substances such as platinum as a catalyst, In 2014, Toyota said it would introduce its Toyota Mirai in Japan for less than $70,000 in 2015. Former European Parliament President Pat Cox estimates that Toyota will initially lose about $100,000 on each Mirai sold.
Freezing conditions
The problems in early fuel-cell designs at low temperatures concerning range and cold start capabilities have been addressed so that they “cannot be seen as show-stoppers anymore”. Users in 2014 said that their fuel cell vehicles perform flawlessly in temperatures below zero, even with the heaters blasting, without significantly reducing range. Studies using neutron radiography on unassisted cold-start indicate ice formation in the cathode, three stages in cold start and Nafion ionic conductivity. A parameter, defined as coulomb of charge, was also defined to measure cold start capability.
Service life
The service life of fuel cells is comparable to that of other vehicles. PEM service life is 7,300 hours under cycling conditions.
Hydrogen
Hydrogen does not come as a pre-existing source of energy like fossil fuels but is first produced and then stored as a carrier, much like a battery. A suggested benefit of large-scale deployment of hydrogen vehicles is that it could lead to decreased emissions of greenhouse gasses and ozone precursors. However, as of 2014, 95% of hydrogen is made from methane. It can be produced using renewable sources, but that is an expensive process. Integrated wind-to-hydrogen (power to gas) plants, using electrolysis of water, are exploring technologies to deliver costs low enough, and quantities great enough, to compete with traditional energy sources.
According to Ford Motor Company, “when FCVs are run on hydrogen reformed from natural gas using this process, they do not provide significant environmental benefits on a well-to-wheels basis (due to GHG emissions from the natural gas reformation process).” While methods of hydrogen production that do not use fossil fuel would be more sustainable, currently renewable energy represents only a small percentage of energy generated, and power produced from renewable sources can be used in electric vehicles and for non-vehicle applications.
The challenges facing the use of hydrogen in vehicles include production, storage, transport, and distribution. The well-to-wheel efficiency for hydrogen is less than 25%. More recent analyses confirm this.
Production
The molecular hydrogen needed as an onboard fuel for hydrogen vehicles can be obtained through many thermochemical methods utilizing natural gas, coal (by a process known as coal gasification), liquefied petroleum gas, biomass (biomass gasification), by a process called thermolysis, or as a microbial waste product called biohydrogen or Biological hydrogen production. 95% of hydrogen is produced using natural gas, and 85% of hydrogen produced is used to remove sulfur from gasoline. Hydrogen can also be produced from water by electrolysis at working efficiencies in the 50–60% range for the smaller electrolysers and around 65–70% for the larger plants. Hydrogen can also be made by chemical reduction using chemical hydrides or aluminum. Current technologies for manufacturing hydrogen use energy in various forms, totaling between 25 and 50 percent of the higher heating value of the hydrogen fuel, used to produce, compress or liquefy, and transmit the hydrogen by pipeline or truck.,
Environmental consequences of the production of hydrogen from fossil energy resources include the emission of greenhouse gasses, a consequence that would also result from the on-board reforming of methanol into hydrogen. Analyses comparing the environmental consequences of hydrogen production and use in fuel-cell vehicles to the refining of petroleum and combustion in conventional automobile engines do not agree on whether a net reduction of ozone and greenhouse gasses would result. Hydrogen production using renewable energy resources would not create such emissions, but the scale of renewable energy production would need to be expanded to be used in producing hydrogen for a significant part of transportation needs. As of 2016, 14.9 percent of U.S. electricity was produced from renewable sources. In a few countries, renewable sources are being used more widely to produce energy and hydrogen. For example, Iceland is using geothermal power to produce hydrogen, and Denmark is using wind.
Storage
Compressed hydrogen in hydrogen tanks at 350 bar (5,000 psi) and 700 bar (10,000 psi) is used for hydrogen tank systems in vehicles, based on type IV carbon-composite technology.
Hydrogen has a very low volumetric energy density at ambient conditions, equal to about one-third that of methane. Even when the fuel is stored as liquid hydrogen in a cryogenic tank or in a compressed hydrogen storage tank, the volumetric energy density (megajoules per liter) is small relative to that of gasoline. Hydrogen has three times higher specific energy by mass compared to gasoline (143 MJ/kg versus 46.9 MJ/kg). In 2011, scientists at Los Alamos National Laboratory and University of Alabama, working with the U.S. Department of Energy, found a single-stage method for recharging ammonia borane, a hydrogen storage compound. In 2018, researchers at CSIRO in Australia powered a Toyota Mirai and Hyundai Nexo with hydrogen separated from ammonia using a membrane technology. Ammonia is easier to transport safely in tankers than pure hydrogen.
Infrastructure
The hydrogen infrastructure for consists of hydrogen-equipped filling stations, which are supplied with hydrogen via compressed hydrogen tube trailers, liquid hydrogen tank trucks or dedicated onsite production, and some industrial hydrogen pipeline transport. The distribution of hydrogen fuel for vehicles throughout the U.S. would require new hydrogen stations that would cost between 20 billion dollars in the US, (4.6 billion in the EU). and half trillion dollars in the US.
As of 2018, there were 40 publicly accessible hydrogen refueling stations in the US, most which are in located in California (compared with 19,000 electric charging stations). By 2017, there were 91 hydrogen fueling stations in Japan.
Codes and standards
Hydrogen codes and standards, as well as codes and technical standards for hydrogen safety and the storage of hydrogen, have been identified as an institutional barrier to deploying hydrogen technologies and developing a hydrogen economy. To enable the commercialization of hydrogen in consumer products, new codes and standards must be developed and adopted by federal, state and local governments.
Safety
Hydrogen powered cars are no more dangerous than petrol or gas powered vehicles. Hydrogen is a very volatile gas because of its low density. It evaporates very quickly outdoors. Provide adequate ventilation in confined spaces as it is flammable over a wide range of 4-75 vol.% (Gasoline: 0.6-8 vol.%). Oxygen / hydrogen mixtures containing less than 10.5% hydrogen by volume are heavier than air and sink to the bottom. The segregation does not take place directly, so that ignitability is maintained until it falls below the 4% by volume limit. When handling hydrogen, safety regulations and ventilation systems must take this behavior into account.
Gasoline is a liquid that slowly evaporates. The inflammable gasoline vapors are heavier than air and remain on the ground for a longer time, and the period of time in which they can ignite is longer.
If hydrogen is released in closed rooms, there is an increased risk of explosion, eg. B. in garages or tunnels. Here is to ensure increased ventilation and possibly additional security measures.
The detonation limit of hydrogen is at a concentration of 18%. Gasoline explodes much earlier, already at a concentration of 1.1%. In order for an explosion or a fire to occur at all, in both cases, a fuel-air mixture that has arisen must first be ignited. In the case of hydrogen, this requires a lower energy of 0.02 mJ than gasoline (gasoline: 0.24 mJ), but in practice it does not matter because even the energy of an electric spark is enough to produce gasoline fumes ignite.
Gasoline has a significantly lower ignition temperature (220-280 ° C) than hydrogen (585 ° C), making it easier to ignite on hot surfaces such as the exhaust manifold or catalyst.
After ignition, hydrogen burns at a higher burning rate than gasoline. The flame moves steeply upwards with a small diameter, if the leak is at the top of the tank.
A hydrogen flame has less heat radiation than a gasoline flame. In addition to a hydrogen flame, it is therefore less hot than next to a gasoline flame – the advantage is that adjacent items such. B. Car seats are not so easy to catch fire. Also, people who are near the flame are less likely to suffer burns. However, the hydrogen flame is barely visible. Therefore, there is a risk of involuntary involvement.
The pressure tanks used today hold (as opposed to gasoline tanks) even severe accidents without prejudice. Hydrogen vehicles with pressure tanks can be easily parked in parking garages and underground garages. There is no legal provision restricting this. In contrast, vehicles with liquid hydrogen must not be stored in closed rooms, as the outgassing can cause explosive gas accumulation.
The main problem with hydrogen storage is leaks. Hydrogen tanks and piping must be due to the z. As natural gas or propane / butane smaller molecular diameter be sealed much better. Some materials are unsuitable because they are permeable to hydrogen. Leaks not only lead to high transport losses, but create a safety hazard when gas accumulates and forms a hydrogen-air mixture. That is why hydrogen tanks and pipes are made of special plastics that largely prevent diffusion . Such systems must be approved by the TÜV. It is advantageous that hydrogen escapes upwards because of its low density and does not collect in depressions, in contrast to gasoline vapors, propane or butane.
Comparison with other types of alternative fuel vehicle
Hydrogen vehicles compete with various proposed alternatives to the modern fossil fuel powered vehicle infrastructure.
Plug-in hybrids
Plug-in hybrid electric vehicles, or PHEVs, are hybrid vehicles that can be plugged into the electric grid and contain an electric motor and also an internal combustion engine. The PHEV concept augments standard hybrid electric vehicles with the ability to recharge their batteries from an external source, enabling increased use of the vehicle’s electric motors while reducing their reliance on internal combustion engines. The infrastructure required to charge PHEVs is already in place, and transmission of power from grid to car is about 93% efficient. This, however, is not the only energy loss in transferring power from grid to wheels. AC/DC conversion must take place from the grids AC supply to the PHEV’s DC. This is roughly 98% efficient. The battery then must be charged. As of 2007, the Lithium iron phosphate battery was between 80-90% efficient in charging/discharging. The battery needs to be cooled; the GM Volt’s battery has 4 coolers and two radiators. As of 2009, “the total well-to-wheels efficiency with which a hydrogen fuel cell vehicle might utilize renewable electricity is roughly 20% (although that number could rise to 25% or a little higher with the kind of multiple technology breakthroughs required to enable a hydrogen economy). The well-to-wheels efficiency of charging an onboard battery and then discharging it to run an electric motor in a PHEV or EV, however, is 80% (and could be higher in the future)—four times more efficient than current hydrogen fuel cell vehicle pathways.” A 2006 article in Scientific American argued that PHEVs, rather than hydrogen vehicles, would become standard in the automobile industry. A December 2009 study at UC Davis found that, over their lifetimes, PHEVs will emit less carbon than current vehicles, while hydrogen cars will emit more carbon than gasoline vehicles.
Natural gas
Internal combustion engine-based compressed natural gas(CNG), HCNG or LNG vehicles (Natural gas vehicles or NGVs) use methane (Natural gas or Biogas) directly as a fuel source. Natural gas has a higher energy density than hydrogen gas. NGVs using biogas are nearly carbon neutral. Unlike hydrogen vehicles, CNG vehicles have been available for many years, and there is sufficient infrastructure to provide both commercial and home refueling stations. Worldwide, there were 14.8 million natural gas vehicles by the end of 2011. The other use for natural gas is in steam reforming which is the common way to produce hydrogen gas for use in electric cars with fuel cells.
All-electric vehicles
A 2008 Technology Review article stated, “Electric cars—and plug-in hybrid cars—have an enormous advantage over hydrogen fuel-cell vehicles in utilizing low-carbon electricity. That is because of the inherent inefficiency of the entire hydrogen fueling process, from generating the hydrogen with that electricity to transporting this diffuse gas long distances, getting the hydrogen in the car, and then running it through a fuel cell—all for the purpose of converting the hydrogen back into electricity to drive the same exact electric motor you’ll find in an electric car.” Thermodynamically, each additional step in the conversion process decreases the overall efficiency of the process.
Source from Wikipedia